
|
|
ENCYCLOPEDIA OF RADIO ELECTRONICS AND ELECTRICAL ENGINEERING Regeneration of galvanic cells and batteries. Encyclopedia of radio electronics and electrical engineering
Encyclopedia of radio electronics and electrical engineering / Chargers, batteries, galvanic cells The idea of restoring discharged galvanic cells, like batteries, is not new. Restore elements using special chargers. It has been practically established that the most common cup manganese-zinc cells and batteries, such as 3336L (KBS-L-0,5), 3336X (KBS-X-0,7), 373, 336, are better than others. manganese-zinc batteries "Krona VTs", BASG and others. The best way to regenerate chemical power supplies is to run an asymmetric alternating current through them, which has a positive DC component. The simplest source of asymmetric current is a half-wave rectifier based on a diode shunted by a resistor. The rectifier is connected to the secondary low-voltage (5-10 V) winding of a step-down transformer powered from the AC mains. However, such a charger has a low efficiency - about 10% and, in addition, the battery being charged can be discharged if the voltage supplying the transformer is accidentally disconnected. Better results can be achieved if a charger is used, made according to the scheme shown in fig. 1. In this device, the secondary winding II feeds two separate rectifiers on diodes D1 and D2, to the outputs of which two rechargeable batteries B1 and B2 are connected.
Capacitors C1 and C2 are connected in parallel with diodes D1 and D2. On fig. 2 shows an oscillogram of the current passing through the battery. The shaded part of the period is the time during which the discharge current pulses flow through the battery.
These impulses, obviously, in a special way affect the course of electrochemical processes in the active materials of galvanic cells. The processes occurring in this case are still insufficiently studied and there is no description of them in the popular literature. In the absence of discharge current pulses (which happens when a capacitor connected in parallel with the diode is disconnected), the regeneration of the elements practically ceased. It has been experimentally established that manganese-zinc galvanic cells are relatively little critical to the magnitude of the constant component and the shape of the negative pulses of the charging current. This allows you to use the charger without additional adjustment of the constant and variable components of the charging current for recovery, various cells and batteries. The ratio of the constant component of the charge current to the effective value of its variable component should be within 5-25. The performance of the charger can be improved by enabling multiple cells to be charged in series. In this case, it is necessary to take into account that in the process of charging e. d.s. elements can increase up to 2-2,1.v. Based on this and knowing the voltage on the secondary winding of the transformer, the number of simultaneously charged elements is determined. It is more convenient to connect batteries of type 3336L to the charger through an incandescent bulb 2,5v X 0,2a, which plays the role of a barter and at the same time serves as an indicator of the degree of charge. As the electric charge of the battery is restored, the glow of the light bulb decreases. Elements of the "Mars" type (373) must be connected without a light bulb, since the constant component of the charging current of such an element should be 200-400 mA. Elements 336 are connected in groups of three, connected in series. Charging conditions are the same as for batteries type 3336. Charging current for cells 312, 316 should be 30-60 mA. It is possible to simultaneously charge large groups of 3336L (3336X) batteries directly from the mains (without a transformer) through two D226B diodes connected in series, in parallel with which a 0,5 μF capacitor with an operating voltage of 600 V is connected. The charger can be made on the basis of the Molodist electric razor transformer, which has two secondary windings with a voltage of 7,5 V. It is also convenient to use the 6,3 V filament voltage of any mains tube radio. Naturally, one or another solution is chosen depending on the required maximum charging current, determined by the type of elements being restored. From the same proceed, choosing rectifier diodes.
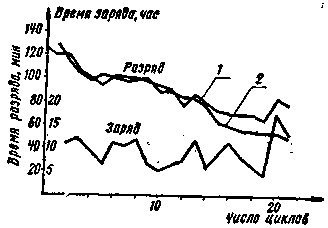
In order to evaluate the effectiveness of this method for the recovery of galvanic cells and batteries, in Fig. Figure 3 shows the discharge voltage graphs for two 3336L batteries with a load resistance Rн=10 ohm. The solid lines show the discharge curves of new batteries, and the dotted lines show the curves after twenty complete discharge-charge cycles. Thus, the performance of the batteries after twenty times of use is still quite satisfactory. How many discharge-charge cycles can galvanic cells and batteries withstand? Obviously, this is highly dependent on operating conditions, storage periods and other factors. On fig. 4 shows the change in the discharge time to the load Rn=10 ohm of two 3336L batteries (curves 1 and 2) during 21 discharge-charge cycles. The batteries were discharged to a voltage of at least 2,1 V, the charge mode of both batteries was the same. During the specified time of battery operation, the discharge time decreased from 120-130 minutes to 50-80 minutes, that is, almost twice.
The same reduction in capacity is allowed by the technical specifications at the end of the specified maximum storage period. In practice, it is possible to restore cells and batteries until their zinc cups are completely destroyed or the electrolyte dries up. It has been established that elements that are intensively discharged to a powerful load (for example, in flashlights, in power supplies for electric shavers) withstand more cycles. Cells and batteries should not be discharged below 0,7 V per cell. The recoverability of elements 373 is relatively worse, since after 3-6 cycles their capacity decreases sharply. The required duration of the charge can be concluded using the graph; shown in fig. 4. With an increase in the charge time over 5 hours, the restored battery capacity increases, on average, very slightly. Therefore, we can assume that at the indicated values of the charging current, the minimum recovery time is 4-6 hours, and manganese-zinc cells do not have obvious signs of the end of the charge and are insensitive to overcharging. The use of asymmetric current is also useful for charging and forming accumulators and storage batteries. This question, however, still needs to be tested in practice and may open up interesting new possibilities for batteries. Author: I. Alimov, Amur Region; Publication: N. Bolshakov, rf.atnn.ru
Machine for thinning flowers in gardens
02.05.2024 Advanced Infrared Microscope
02.05.2024 Air trap for insects
01.05.2024
▪ The most dangerous air pollution ▪ High-speed Internet between the Earth and the Moon
▪ site section Chargers, accumulators, batteries. Article selection ▪ article This mystery is great. Popular expression ▪ article How many animals can live on a single tree? Detailed answer ▪ article Cleaner of industrial and service premises. Standard instruction on labor protection ▪ article Cement - putty for the aquarium. Simple recipes and tips ▪ article A simple battery charger. Encyclopedia of radio electronics and electrical engineering
Comments on the article: Oleg In those years when this article was written, it was very important because it was extremely difficult to buy these elements. I tried to do it. The result was good. I recommend. And today it's easier to buy a new battery Moysha To recharge the batteries for "every day" any device with a charge current of 1-5 mA will do. I use a half-wave rectifier with a resistor, for AA-type energizer batteries. I use batteries in a tourist navigator for mushroom picking. With constant night resuscitation, it is enough for the whole season.
Home page | Library | Articles | Website map | Site Reviews www.diagram.com.ua |






 Arabic
Arabic Bengali
Bengali Chinese
Chinese English
English French
French German
German Hebrew
Hebrew Hindi
Hindi Italian
Italian Japanese
Japanese Korean
Korean Malay
Malay Polish
Polish Portuguese
Portuguese Spanish
Spanish Turkish
Turkish Ukrainian
Ukrainian Vietnamese
Vietnamese



 Leave your comment on this article:
Leave your comment on this article: