
|
|
ENCYCLOPEDIA OF RADIO ELECTRONICS AND ELECTRICAL ENGINEERING Rules for charging alkaline nickel-cadmium and nickel-metal hydrate batteries. Encyclopedia of radio electronics and electrical engineering
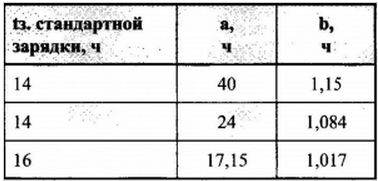
Encyclopedia of radio electronics and electrical engineering / Chargers, batteries, galvanic cells Sealed alkaline nickel-cadmium (NiCd) and nickel-metal hydrate (NiMH) batteries are increasingly used not only in industrial devices, but also in household appliances, replacing AA and AAA batteries. But these batteries, like any other, eventually "empty" and require charging. For recharging, as recommended by the operating instructions and the marking of the batteries themselves, it is necessary to pass a current through them Ist \u0,1d 12 mA from the numerical value of the nominal capacity C for 16-XNUMX hours. But what if there is no suitable current or "smart" electronic charger, and the available simple transformerless charge current differs from the battery rating? It's hard to estimate the charging time exactly. And if you determine it by eye, then if the batteries are not fully charged, the capabilities of the batteries will be limited, and if they are excessive, the service life will be reduced and they may even explode! Therefore, so that when using a non-native charger, in which the charge current Iz differs from the standard one (0,1 C), it is proposed to determine the charging time tc using a special formula. The following main points were taken as a basis for its conclusion: 1. Charging time ts (hour) is inversely proportional to the charging current I (A or mA) and directly proportional to the battery capacity C (Ah or mAh). 2. Charging current should not be more than allowed for "fast charging" Ib. If it is not specified, then take it equal to 0,25 of the numerical value of the capacitance. (Before using the existing charger, it is necessary to measure the "de facto" current - it may differ from the passport values). 3. When servicing the battery, the main part of the current goes directly to charging - electrochemical processes, and the other part is spent on auxiliary work and related phenomena: ion movement, heating and other losses. From the above it follows that the theoretical "ideal" charging time can be determined by the formula: ts=C/I (1). Taking into account the losses, we introduce the correction factor K ts=K*C/I (2), where K \u1,2d 0,25 with "fast charging" (Ib \u1,4d 1,6 C) and K \u0,1d XNUMX - XNUMX with standard (Ist \uXNUMXd XNUMX C). These correction factor values are derived from the characteristics of most batteries. Formula (2) by itself is already sufficiently accurate and acceptable for practical use. But the values of K can be refined. Let us express K through a linear dependence of the type y = ax + b (3). Then the general formula will take the form: tz \u4d (C / I.a + b) * C / I (XNUMX). Knowing the capacity and recommended charging currents for standard and "fast" charging and the corresponding charging times, it is possible to calculate the coefficients a and b for a particular battery. For example: we have a nickel-cadmium battery with a capacity of 300 mA * h, with a standard current of 30 mA, the time tc = 16 hours, with "fast charging": 16 = 75 mA and tc = 5 hours. Substituting these data into formula (4), we obtain two linear equations with two unknowns. After solving the system, we get: time factor a = 17,15 h, dimensionless coefficient b = 1,017. Knowing these coefficients, it is easy to calculate by formula (4) the charging time of any batteries with this charger. For example, if the available charger current is 50 mA (0,17 C), then the estimated charge time is 8,2 hours (8 hours 12 minutes). For practical use, below is a table for selecting the coefficients a and b, depending on the standard charging time indicated on the battery.
Note. Charging time should not exceed 20 - 24 hours. With a small Iz, when the value of tz is measured in tens of hours, it is probably better to use a single simplified formula tz=tz.cm.C/10 Iz Author: A.Ignatiev
Machine for thinning flowers in gardens
02.05.2024 Advanced Infrared Microscope
02.05.2024 Air trap for insects
01.05.2024
▪ Drone that forms clouds and causes precipitation ▪ Icis smart glasses from Laforge Optical ▪ High sensitivity 20 MP image sensor from Toshiba ▪ The retina does not change with age ▪ Boys to the left, girls to the right
▪ section of the site For a beginner radio amateur. Article selection ▪ article Encounters with predatory animals. Basics of safe life ▪ article When were the first coins made? Detailed answer ▪ article Working on a round-laminating machine. Standard instruction on labor protection ▪ article Maori proverbs and sayings. Large selection
Home page | Library | Articles | Website map | Site Reviews www.diagram.com.ua |






 Arabic
Arabic Bengali
Bengali Chinese
Chinese English
English French
French German
German Hebrew
Hebrew Hindi
Hindi Italian
Italian Japanese
Japanese Korean
Korean Malay
Malay Polish
Polish Portuguese
Portuguese Spanish
Spanish Turkish
Turkish Ukrainian
Ukrainian Vietnamese
Vietnamese
 Leave your comment on this article:
Leave your comment on this article: